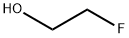Synthesis
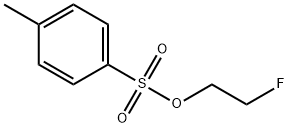
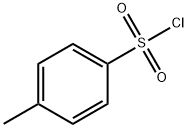
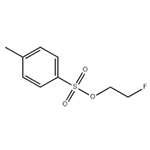
2-Fluoroethanol (1.00 mL, 16.9 mmol), p-toluenesulfonyl chloride (3.85 g, 20.2 mmol), N-methylmorpholine (9.30 mL, 84.5 mmol), and dichloromethane (50 mL) were added to the reaction vessel, and the reaction was stirred for 17 hours at room temperature. After completion of the reaction, water was added to the reaction system and extracted with ethyl acetate three times to combine the organic layers. The organic layer was washed with saturated brine and dried over anhydrous magnesium sulfate. After concentration under reduced pressure, the residue was purified by fast silica gel column chromatography (eluent: ethyl acetate/hexane=19:81→40:60) to afford fluoroethyl p-toluenesulfonate (3.79 g, 17.2 mmol, yield >100%) as a pale yellow liquid.1H NMR (600 MHz, CDCl3): δ 7.81 (d, J=8.4 Hz, 2H), δ 7.36 (d, J=8.4 Hz, 2H), 4.63-4.60 (m, 1H), 4.55-4.52 (m, 1H), 4.31-4.27 (m, 1H), 4.26-4.23 (m, 1H), 2.46 (s, 3H). lRMS (EI) Calculated value (C9H11F3O): 218.0, measured value: 218.0.
References
[1] Patent: US2018/30074, 2018, A1. Location in patent: Paragraph 0188-0191
[2] Journal of Labelled Compounds and Radiopharmaceuticals, 2008, vol. 51, # 7, p. 286 - 292
[3] Patent: US2013/177501, 2013, A1. Location in patent: Paragraph 0168
[4] Patent: US9168317, 2015, B2. Location in patent: Page/Page column 25; 26
[5] Patent: US9314541, 2016, B2. Location in patent: Page/Page column 26