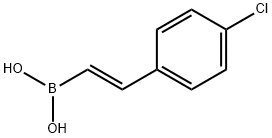
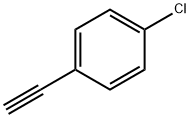
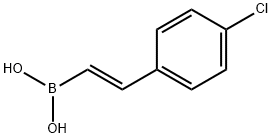
Uses
Reactant involved in:• ;Synthesis of biarylketones and phthalides1• ;Trifluoromethylation2• ;Asymmetrical Michael addition for preparation of chromanes3• ;Cobalt-catalyzed coupling to vinyl nitrogen containing heteroaromatic compounds4• ;1,2 and 1,4-Addition reactions with o-hydroxycinnamaldehydes5• ;Petasis reactions6
Synthesis
General procedure for the synthesis of (E)-(4-chlorophenylvinyl)boronic acid from 4-chlorophenylacetylene and catecholborane: 1-chloro-4-ethynylbenzene (0.50 g, 3.66 mmol, 1.00 eq.) and catecholborane (0.47 mL, 4.39 mmol, 1.20 eq.) were dissolved in tetrahydrofuran (THF, 1.4 mL) and the mixture was refluxed for for 18 hours. After completion of the reaction, the solvent was removed by evaporation and water (1 mL) was added. The suspension was stirred vigorously for 4 h at room temperature. The solid was collected by filtration and recrystallized from water. It was filtered again to give (E)-(4-chlorophenylvinyl)boronic acid (652.9 mg, 3.62 mmol, 99% yield) and dried under vacuum. The product was characterized by 1H NMR (400 MHz, DMSO-d6) and 13C NMR (101 MHz, DMSO) with the following data: 1H NMR (DMSO-d6) δ 7.83 (s, 2H), 7.50 (d, J = 8.4 Hz, 2H), 7.45-7.39 (m, 2H), 7.24 (d, J = 18.4 Hz. 1H), 6.14 (d, J = 18.3 Hz, 1H); 13C NMR (DMSO) δ 145.7, 144.8, 137.0, 133.3, 129.2 (2C), 128.7 (2C).