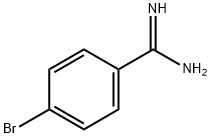
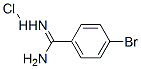
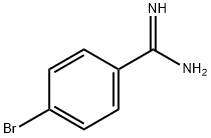
Synthesis
The general procedure for the synthesis of 4-bromobenzamidine hydrochloride from 4-bromobenzamidine hydrochloride was as follows: first, 4-bromobenzamidine hydrochloride (6a, 707 mg) was treated with 5 M aqueous sodium hydroxide (8 mL), followed by extraction with chloroform. The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give a colorless solid (472 mg). Next, the resulting solid was dissolved in ethanol (5 mL), ethyl propargylate (0.27 mL) was added and the reaction mixture was heated to 60 °C. Then, a solution of potassium hydroxide (175 mg) in ethanol (4 mL) was added dropwise over 15 minutes. The reaction mixture was further heated to 80 °C and stirred at this temperature for 6 hours. Upon completion of the reaction, the mixture was cooled to room temperature, acidified by addition of 1 M hydrochloric acid (10 mL) and the precipitate was collected by filtration. The precipitate was washed with water and dried under vacuum at 50 °C overnight to give 7 g (267 mg, 35% yield) of the target product as a light brown solid. The product was characterized by 1H NMR (DMSO-d6): δ 6.25-5.50 (1H, m), 7.74 (2H, d, J = 8.3 Hz), 7.95-8.25 (3H, m), 12.50-13.20 (1H, br); FAB-MS m/z 251,253 [(M + H)+].
References
[1] Bioorganic and Medicinal Chemistry, 2012, vol. 20, # 7, p. 2369 - 2375
[2] Patent: EP2862856, 2015, A1. Location in patent: Paragraph 0651; 0652
[3] Tetrahedron Letters, 2018, vol. 59, # 4, p. 361 - 364