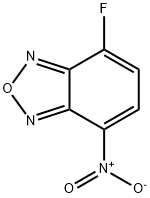
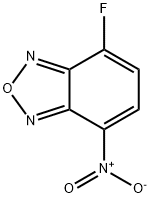
Chemical Properties
BROWN CRYSTALLINE CHUNKS
Uses
4-Chloro-7-nitrobenzofurazan (NBD-chloride) was used in the following studies, Synthesis of fluorescent phospholipid-derivative, NBD-didecanoylphosphatidylethanolamine, Synthesis of functionalized hydroxynaphthofurazan, Spectrophotometric and spectrofluorometric determination of clemastine hydrogen fumarate, loratadine, losartan potassium and ramipril in pharmaceutical formulations. Synthesis of 7-nitrobenzofurazan (NBD)-labeled maleimide, via Diels-Alder reaction. NBD-Cl is widely used to label peptides, proteins, drugs and other biomolecules. It is also a popular derivatizing reagent for HPLC analysis
Uses
A fluorescence reagent
Synthesis
General procedure for the synthesis of 4-fluoro-7-nitro-2,1,3-benzoxadiazole from 4-fluoro-2,1,3-benzoxadiazole: 4-fluoro-2,1,3-benzoxadiazole (1.00 g, 7.24 mmol) was dissolved in concentrated H2SO4 (8 mL) and cooled to -10 °C. The nitration mixture (H2SO4:HNO3, 3:1, 2.40 mL) was added slowly and dropwise with stirring. The reaction temperature was maintained at -10 °C and stirring was continued for 1 hour. The progress of the reaction was monitored by TLC and LCMS. Upon completion of the reaction, the reaction mixture was quenched with H2O (250 mL) and extracted with EtOAc (2 x 250 mL). The organic layers were combined, dried with anhydrous Na2SO4 and concentrated under reduced pressure. The crude product was purified by fast column chromatography (eluent: 0 to 10% EtOAc gradient in hexane) to afford 4-fluoro-7-nitro-2,1,3-benzoxadiazole (0.39 g, yellow solid) in 30% yield.1H NMR (400 MHz, DMSO-d6) δ 7.70 (d, J=8.40 Hz, 1H), 8.77-8.80 (m 1H).
Purification Methods
Purify it by repeated recrystallisation from pet ether (b 40-60o). On treatment with MeONa in MeOH it gives 4-methoxy-7-nitrobenzo-2-oxa-1,3-diazole m 115-116o. [Nunno et al. J Chem Soc (C) 1433 1970.] It is a very good fluorophore for amino acids [Imai & Watanabe Analyt Chim Acta 130 377 1981], as it reacts with primary and secondary amines to form fluorescent adducts with 470nm and 530nm. It gives a glycine derivative with m 185-187o [Miyano et al. Anal Chim ex em Acta 170 81 1985]. 5-Fluorouracil (5 -fluoropyrimidinedi -2,4 -[1 H ,3 H ]-one) [51 -21 -8] M 130.1, m 282 -2 8 3o(dec), 282-286o(dec), pK 1 8.04, pK 2 13.0. Recrystallise it from H2O or MeOH/Et2O and sublime it at 190-200o/0.1mm or 210-230o/0.5mm. UV: at 265-266nm ( 7070). [Barton et al. J Org max Chem 37 329 1972, Duschinsky & Pleven J Am Chem Soc 79 4559 1957, Beilstein 24 III/IV 1229.]
References
[1] Journal of the American Chemical Society, 2016, vol. 138, # 48, p. 15596 - 15604
[2] Patent: WO2018/122232, 2018, A1. Location in patent: Page/Page column 213
[3] Tetrahedron Letters, 2011, vol. 52, # 20, p. 2533 - 2535