Synthesis
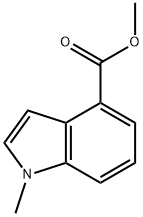
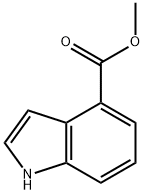
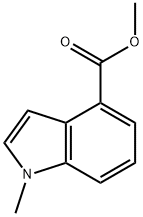
To a 40 mL reaction vial was added a solution of methyl 1H-indole-4-carboxylate (400 mg, 2.28 mmol, 1.00 equiv) in N,N-dimethylformamide (10 mL). Sodium hydride (275 mg, 6.88 mmol, 3.00 equiv, 60% dispersed in mineral oil) was added in batches at 0 °C. After keeping stirring at 0 °C for 0.5 h, iodomethane (1.298 g, 9.14 mmol, 4.00 eq.) was slowly added dropwise at the same temperature. The reaction mixture was warmed to 25 °C and stirred for 15 hours. After completion of the reaction, the mixture was poured into ice water (50 mL) and extracted with ethyl acetate (50 mL x 3). The organic phases were combined, washed with saturated saline (100 mL), dried over anhydrous sodium sulfate, filtered and concentrated. Methyl 1-methyl-4-indolecarboxylate (429 mg, 99%) was obtained as a red oil after concentration under reduced pressure. Mass spectrum (electrospray ionization, m/z): [M+H]+ 190.
References
[1] Patent: WO2017/205296, 2017, A1. Location in patent: Paragraph 78
[2] Patent: US2009/298809, 2009, A1. Location in patent: Page/Page column 17
[3] Heterocycles, 2006, vol. 67, # 2, p. 643 - 653