Synthesis
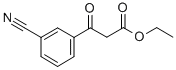
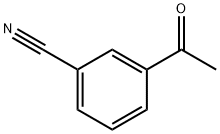
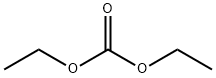
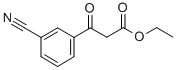
Part A. Preparation of ethyl 3-(3-cyanophenyl)-3-oxopropanoate. To a hexane-washed suspension of sodium hydride (1.2 g, 60% mineral oil suspension, 30.3 mmol) in 40 mL of tetrahydrofuran, diethyl carbonate (3.7 mL, 30.3 mmol) and 3-acetylbenzonitrile (2.2 g, 15.2 mmol) were added sequentially. The resulting suspension was stirred at 65 °C for 1 h and then cooled to room temperature. 40 mL of 10% HCl aqueous solution was added and the reaction mixture was diluted with ethyl acetate and layered. The organic layer was washed with brine, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to afford 3.2 g (96% yield) of ethyl 3-(4-cyanophenyl)-oxopropanoate, which was pure enough to be used directly in subsequent reactions without further purification. Mass spectrum (NH3-Cl) m/z: 218.3 ([M+H]+).
References
[1] Journal of Medicinal Chemistry, 2006, vol. 49, # 6, p. 1910 - 1915
[2] Bioorganic and Medicinal Chemistry Letters, 2001, vol. 11, # 5, p. 641 - 645
[3] Patent: WO2011/46774, 2011, A1. Location in patent: Page/Page column 16
[4] Patent: EP946528, 2003, B1
[5] Patent: US6187797, 2001, B1