Synthesis
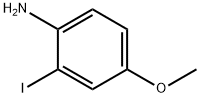
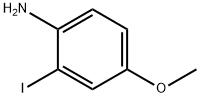
The general procedure for the synthesis of 2-iodo-4-methoxyaniline from tert-butyl (2-iodo-4-methoxyphenyl)carbamate was as follows: tert-butyl (2-iodo-4-methoxyphenyl)carbamate (3.5 g, 10.0 mmol) was slowly added to a solution of (2-iodo-4-methoxyphenyl)carbamate in dichloromethane (50 mL) at 0 °C with trifluoroacetic acid (8.9 mL, 120.0 mmol). The reaction mixture was stirred at room temperature for 2 hours. Upon completion of the reaction, the mixture was cooled to 0 °C and the reaction was quenched with 2N sodium hydroxide solution (95 mL), followed by adjusting the pH to 8 with 2N hydrochloric acid (70 mL).The organic and aqueous layers were separated, and the aqueous layer was extracted with dichloromethane (2 x 70 mL). All organic layers were combined, dried with anhydrous sodium sulfate and concentrated under reduced pressure. The residue was purified by fast column chromatography (silica gel, n-hexane/ethyl acetate = 7:1) to give 2-iodo-4-methoxyaniline (2.12 g, 85% yield) as an orange oil.
References
[1] Organic and Biomolecular Chemistry, 2016, vol. 14, # 6, p. 2127 - 2133
[2] Chemical Communications, 2001, # 24, p. 2732 - 2733
[3] Organic and Biomolecular Chemistry, 2003, vol. 1, # 1, p. 117 - 122
[4] Organometallics, 2013, vol. 32, # 2, p. 682 - 690
[5] Tetrahedron, 2013, vol. 69, # 45, p. 9481 - 9493