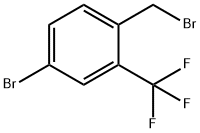
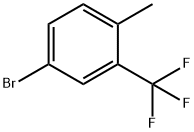
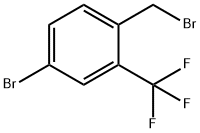
Synthesis
General procedure for the synthesis of 4-bromo-1-(bromomethyl)-2-(trifluoromethyl)benzene from 5-bromo-2-methyltrifluorotoluene: 5-bromo-2-methyltrifluorotoluene (25 g, 104.59 mmol), N-bromosuccinimide (18.62 g, 104.59 mmol) and benzoyl peroxide (1.27 g, 5.23 mmol) were reacted in a A mixture in carbon tetrachloride (35 mL) was heated and reacted at 90 °C for 4 hours. Upon completion of the reaction, the mixture was cooled to 0 °C and filtered through a glass sand core funnel and the solids were washed with dichloromethane. The filtrates were combined and concentrated and subsequently purified by fast column chromatography (eluent: hexane/ethyl acetate, 0% to 5% ethyl acetate gradient) to give a clear oily product, which was crystallized after standing (33.26 g, 100% yield). The structure of the product was confirmed by 1H NMR (400 MHz, CDCl3): δ 4.57 (s, 2H), 7.46 (d, J = 8.3 Hz, 1H), 7.67 (d, J = 8.3 Hz, 1H), 7.78 (s, 1H).
References
[1] Patent: WO2006/18725, 2006, A1. Location in patent: Page/Page column 208
[2] Journal of Labelled Compounds and Radiopharmaceuticals, 2015, vol. 58, # 5, p. 188 - 195
[3] Patent: WO2008/148867, 2008, A2. Location in patent: Page/Page column 37
[4] Patent: WO2009/23964, 2009, A1. Location in patent: Page/Page column 24
[5] Patent: WO2006/46593, 2006, A1. Location in patent: Page/Page column 235