Synthesis
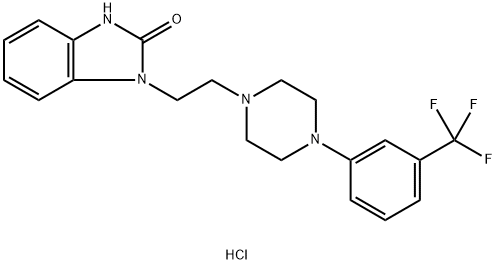
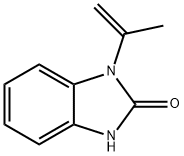
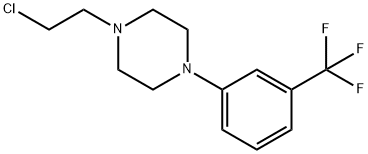
The one-pot reaction of benzaldehyde, N-phenylhydroxylamine and trimethylcyanosilane facilitated by iodobenzene acetate first builds the important backbone benzimidazolone structure of flubanserin, and then reacts with 1,2-dibromoethane to give intermediate 8, which is then hydrolyzed in concentrated hydrochloric acid to give intermediate 9, and then finally reacts with piperazine hydrochloride10 to give flubanserin in a total yield of 29%.11 The formation of salts of flubanserin with hydrochloric acid gives flubanserin hydrochloride. Flubanserin hydrochloride is formed by salting flubanserin with hydrochloric acid. This method is characterized by low cost and easy availability of raw materials, few by-products and high yield.