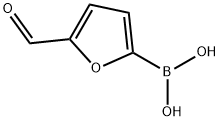
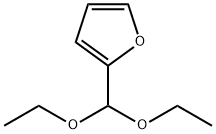
Chemical Properties
beige to brown powder
Uses
As a bifunctional reagent used in the synthesis of π -extended heteroarylfuran systems. Reactant involved in Suzuki coupling for synthesis of stable dye-sensitized solar cells. Reactant involved in synthesis of biologically active molecules including, heteroarylation for the synthesis of HIF-1 inhibitors, disalicylic acid-furanyl derivatives to inhibit ephrin binding, HIV-1 integrase inhibitors, epidermal growth factor receptor inhibitors.
Synthesis
Under nitrogen protection, 20.16 g (0.118 mol) of 2-(diethoxymethyl)furan, 33.4 g (0.177 mol) of triisopropyl borate, and 40 mL of anhydrous tetrahydrofuran (THF) were added to a dry 500 mL three-necked flask equipped with a mechanical stirrer, an internal thermometer, and a dosing funnel. The water content of the reaction mixture was determined by Karl-Fischer titration to ensure that it was less than 800 μg/mL. the reaction mixture was cooled to an internal temperature of -10 °C. The reaction mixture was then purified to a temperature of -10 °C. The water content of the reaction mixture was determined by titration. With the temperature maintained at -10 °C to 0 °C, 84 mL (25 wt%, 1.84 M THF solution containing heptane and ethylbenzene from Chemmetal, content determined by titration, 1.3 equivalents) of LDA solution was slowly added through the addition funnel over a period of 1 hour. Upon completion of addition, the reaction mixture was transferred via cannula to a pre-cooled aqueous hydrochloric acid solution (prepared by mixing 33 mL of concentrated hydrochloric acid and 55 mL of water). The reaction temperature was controlled not to exceed 30 °C. The resulting tan slurry of 5-formylfuran-2-boronic acid was cooled to 0°C and filtered. The filter cake was washed twice with 20 mL of cold water to obtain 17.6 g of wet filter cake. The wet filter cake was dried in a vacuum oven at 40 °C to give 12.41 g of an off-white product, 5-formylfuran-2-boronic acid, in a total yield of 75%. The amount of unreacted furfural in the crude product was determined to be less than 0.1% by analysis.
References
[1] Patent: EP1403271, 2004, A1. Location in patent: Page 3, 4
[2] Organic and Biomolecular Chemistry, 2003, vol. 1, # 9, p. 1447 - 1449