Uses
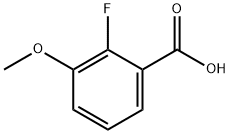
2-Fluoro-3-methoxybenzoic acid (2FMBA) is an intramolecular lithium compound that has been shown to be a potent inhibitor of dihydrofolate reductase. Experiments have shown that 2FMBA can inhibit bacterial growth, bind to DNA, and inhibit RNA synthesis in bacteria.
Synthesis
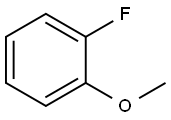
Step 1. Preparation of 2-fluoro-3-methoxybenzoic acid. Potassium tert-butoxide (30.80 g, 274 mmol) was dissolved in anhydrous tetrahydrofuran (300 mL) under nitrogen protection and the mixture was cooled to -78 °C. A hexane solution of 1.6 M n-butyllithium (172 mL, 275 mmol) was slowly added dropwise, and stirring was continued for 15 minutes after completion of the drop. Subsequently, 2-fluoroanisole (31.35 g, 248 mmol) was added and the reaction mixture continued to be stirred at -78 °C for 1.8 hours. Upon completion of the reaction, the mixture was slowly poured into a 2 L Erlenmeyer flask containing dry ice and gradually warmed to room temperature. Water (250 mL) was added to dilute and the organic layer was separated by extraction with ether (160 mL). The aqueous layer was acidified with concentrated hydrochloric acid to pH < 2. A solid was precipitated and collected by filtration to give 2-fluoro-3-methoxybenzoic acid (21.43 g, 51% yield) as a yellow solid. Melting point 155-160 °C. 1H NMR (acetone-d6, 300 MHz) δ 7.46 (ddd, J = 6.0, 1.8, 1.4 Hz, 1H), 7.36 (dt, J = 1.6, 8.1 Hz, 1H), 7.20 (dt, J = 1.4, 8.1 Hz, 1H), 3.92 (s, 3H); 19F NMR (acetone- d6, 300 MHz) δ -134.04 (m). Mass spectrum (ESI+): m/z 171 [M+H]+.