Synthesis
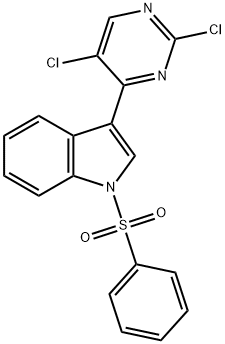
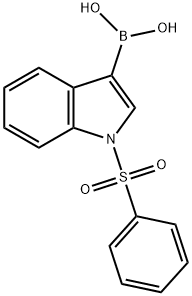
Under nitrogen atmosphere, 2,4,5-trichloropyrimidine (1.25 g) was dissolved in a mixed solvent of anhydrous acetonitrile/water (20 ml/10 ml), followed by the addition of 1-(phenylsulfonyl)-3-indoleboronic acid (2.0 g) and sodium carbonate (1.4 g). The reaction mixture was degassed three times, followed by the addition of tetrakis(triphenylphosphine)palladium(0) (390 mg). After degassing again three times, the mixture was heated to reflux and kept for 90 minutes. Upon completion of the reaction, the precipitate was collected by filtration and washed sequentially with water and ether to afford the target product 3-(2,5-dichloropyrimidin-4-yl)-1-(phenylsulfonyl)-1H-indole as a white solid (2.48 g, 91% yield). The product was characterized by 1H NMR (300 MHz, d6-DMSO): δ 9.00 (1H, s), 8.80 (1H, s), 8.55 (1H, d), 8.15 (2H, d), 8.05-8.02 (1H, s), 7.75 (1H, t), 7.64 (2H, t), 7.52-7.41 (2H, m).
References
[1] Patent: WO2006/38001, 2006, A1. Location in patent: Page/Page column 40
[2] Journal of the American Chemical Society, 2015, vol. 137, # 46, p. 14640 - 14652
[3] Bioorganic and Medicinal Chemistry Letters, 2007, vol. 17, # 12, p. 3463 - 3467