Chemical Properties
White to off-white powder
Synthesis
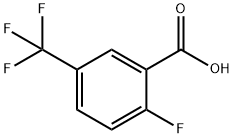
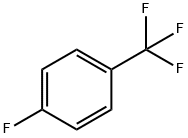
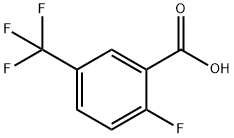
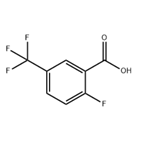
General procedure for the synthesis of 2-fluoro-5-trifluoromethylbenzoic acid from 4-fluorobenzotrifluoride: 99 g (0.6 mol) of 4-fluorobenzotrifluoride was dissolved in 750 ml of tetrahydrofuran at -60 °C and protected by nitrogen, and 256 ml (0.64 mol) of a 2.5 M butyl lithium hexane solution was added slowly dropwise over a controlled period of time. The reaction mixture was stirred continuously at -60 °C to -70 °C for 4 h and subsequently poured into a large excess of dry ice. after 15 min, the excess dry ice was evaporated by external heating and the solvent was removed by rotary evaporator. The residue was dissolved in 500 ml of water, 30 ml of 1N sodium hydroxide solution was added and washed with 2 x 300 ml of ethyl acetate. The aqueous phase was acidified with concentrated hydrochloric acid and then extracted with 2 x 300 ml ethyl acetate. The organic phases were combined and washed sequentially with 500 ml of water and 300 ml of saturated saline, dried over anhydrous sodium sulfate and concentrated by rotary evaporation. The crude product was recrystallized by hexane to give 80 g of white analytically pure crystals with 64% yield and melting point of 205°C. The product was extracted with 500 ml of water and 300 ml of saturated saline. Elemental analysis results (C8H4O2F4): calculated value C 46.17%, H 1.94%; measured value C 46.16%, H 1.89%.
References
[1] Patent: US4705853, 1987, A
[2] Patent: US5304532, 1994, A