Uses
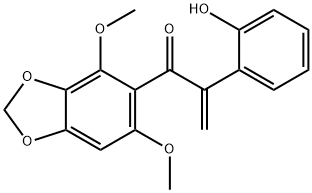
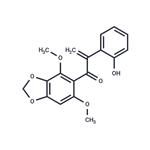
Biliatresone is a natural toxin isolated from Dysphania glomulifera and D. littoralis. Biliatresone, a 1,2-diaryl-2-propenone class of isoflavonoid, produces extrahepatic biliary atresia in a zebrafish model[1].
Definition
ChEBI: Biliatresone is a member of the class of benzodioxoles that is 1,3-benzodioxole which is substituted at positions 5 by a 2-(2-hydroxyphenyl)-3-oxoprop-1-en-3-yl group and at positions 4 and 6 by methoxy groups. A very rare type of isoflavonoid-related 1,2-diaryl-2-propenone found in Dysphania glomulifera (red crumbweed) and D. littoralis, the enone moiety is particularly reactive, undergoing ready Michael addition of water and methanol. Biliatresone has been found to cause extrahepatic biliary atresia (obliteration or discontinuity of the extrahepatic biliary system, resulting in obstruction to bile flow) in a zebrafish model. It has a role as a toxin and a plant metabolite. It is a member of benzodioxoles, an enone, an aromatic ketone, an aromatic ether and a member of phenols.
in vivo
Biliatresone exhibits toxicity at doses of 0.065-1.0 μg/mL with a marked reduction and the lethal dose of Biliatresone in a zebrafish assay is 1 μg/mL[1].
Larvae treated at 5 dpf with low doses of biliatresone [0.0625 μg/ml (0.2 μM) and 0.125 μg/ml (0.4 μM)] shows only subtle gallbladder defects, whereas larvae treated with higher doses has pronounced morphological defects of the gallbladder and extrahepatic ducts[1].
References
[1] Kyung A Koo, et al. Biliatresone, a Reactive Natural Toxin From Dysphania Glomulifera and D. Littoralis: Discovery of the Toxic Moiety 1,2-Diaryl-2-Propenone. Chem Res Toxicol. 2015 Aug 17;28(8):1519-21. DOI:
10.1021/acs.chemrestox.5b00227[2] Kristin Lorent, et al. Identification of a Plant Isoflavonoid That Causes Biliary Atresia. Sci Transl Med. 2015 May 6;7(286):286ra67. DOI:
10.1126/scitranslmed.aaa1652