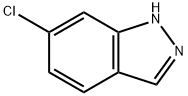
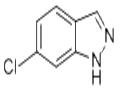
The general procedure for the synthesis of 6-chloro-1H-indazole from 5-chloro-o-toluidine was as follows: aqueous NaNO2 (13.80 g, 0.2 mol, 32.20 mL) was added slowly and dropwise at 0 °C into a reaction system containing 5-chloro-o-toluidine (21.40 g, 0.2 mol), H2O (190 mL), and an aqueous 12N HCl solution (50 mL, 0.6 mol) in a mixed reaction system. The reaction temperature was maintained at 0 °C and filtration was performed after continued stirring for 30 min. To the filtrate, pre-cooled aqueous NaBF4 solution (24.20 g, 0.22 mol dissolved in 90 mL of H2O) was added and stirred at 0 °C for 40 min. The resulting precipitate was collected by filtration, washed sequentially with cold ethanol (50 mL x 3) and cold ether (50 mL x 3), and subsequently concentrated under vacuum to give the diazonium salt (19.19 g) as a yellow solid. The diazonium salt (19.19 g, 0.093 mol) was dissolved in CHCl3 (231 mL) and KOAc (15.15 g, 0.155 mol) was added. The reaction mixture was stirred at room temperature for 4 h, after which the reaction was quenched with water (200 mL) and extracted with CH2Cl2 (70 mL x 3). The organic phases were combined, washed with brine (50 mL × 3), dried over anhydrous Na2SO4 and concentrated under reduced pressure. Finally, the crude product was purified by recrystallization to give 7.51 g of 6-chloro-1H-indazole.