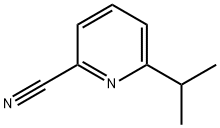
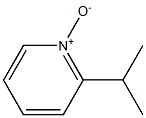
1B: General procedure for the synthesis of 2-cyano-6-isopropylpyridine (105). A mixture of 2-isopropylpyridine-N-oxide (104, 1.33 g, 9.7 mmol) with trimethylcyanosilane (TMS-CN, 1.42 mL, 1.06 g, 11.0 mmol) in 1,2-dichloroethane (40 mL) was stirred at room temperature for 5 min. Subsequently, diethylcarbamoyl chloride (Et2NCOCl, 1.23 mL, 9.7 mmol) was added and stirring was continued at room temperature under inert atmosphere. After 2 days of reaction, 10% aqueous potassium carbonate solution was added and stirring was continued for 10 min. The organic layer was separated and the aqueous layer was extracted twice with 1,2-dichloroethane. The organic layers were combined, washed with brine, dried over anhydrous sodium sulfate and concentrated. The residue was purified by silica gel column chromatography (eluent: hexane/ethyl acetate, 3:1) to afford 6-isopropylpyridinecarbonitrile 1.06 g (74% yield): m/z = 147 (M + H)+.