It is used in organic syntheses and as pharmaceutical intermediates. The best results were obtained with lactams that could be introduced on the 6-bromoimidazo[1,2-a]pyridine. 1,2-dihydro-5-imidazo [1,2-a]pyridin-6-yl-6-methyl-2-oxo-3-pyridinecarbonitrile hydrochloride monohydrate 6 was synthesized in 5 steps from 6-bromoimidazo[1,2-a] pyridine using [2- 14 C] cyanoacetamide as the source of the radiolabel.
(1) Preparation of 6-bromoimidazo[1,2-a]pyridine (4): 2.00 g of 2-amino-5-bromopyridine (3) was dissolved in 35 mL of ethanol, and 2.52 mL of 6.1 mol/L aqueous chloroacetaldehyde solution was added. The reaction mixture was heated to reflux for 3 hours. After completion of the reaction, the reaction mixture was concentrated and saturated aqueous NaHCO3 solution was added to the residue and extracted with ethyl acetate. After conventional post-treatment, 2.30 g of 6-bromoimidazo[1,2-a]pyridine was obtained as a brown powder (yield: 100%).LCMS (ESI) m/z (M + H)+: 368.93.
[1] Patent: US2011/98471, 2011, A1. Location in patent: Page/Page column 56
[2] Patent: US2014/134133, 2014, A1. Location in patent: Paragraph 0399
[3] Patent: WO2012/116237, 2012, A2. Location in patent: Page/Page column 118
[4] Patent: US2015/320727, 2015, A1. Location in patent: Paragraph 0553; 0554
[5] Patent: WO2008/14219, 2008, A2. Location in patent: Page/Page column 44-45
![6-Bromoimidazo[1,2-a]pyridine Structure](/CAS/GIF/6188-23-4.gif)
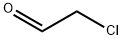

![6-Bromoimidazo[1,2-a]pyridine pictures](/ProductImageEN/2023-12/Small/c723055e-b9c2-4a29-b79e-a26e95a2ede3.jpg)
![6-Bromoimidazo[1,2-a]pyridine pictures](/ProductImageEN/2021-10/Small/0cf4be4d-8f8a-40c6-97b9-525705514b55.jpg)
![6-Bromoimidazo[1,2-a]pyridine pictures](/ProductImageEN/2022-09/Small/ed6da65b-58de-4435-a1d1-79873874e19e.png)