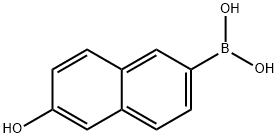
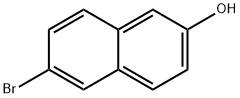
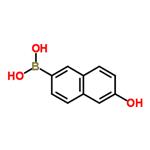
Under argon protection, 5.58 g (25 mmol) of 6-bromo-2-naphthol was dissolved in 125 mL of anhydrous tetrahydrofuran. After cooling the resulting solution to -70°C, 33 mL (55 mmol) of hexane solution of n-butyllithium was added slowly and dropwise over a period of 30 minutes. Maintaining -70°C, the reaction mixture was continued to be stirred for 1.5 hours. Subsequently, 11.5 mL (50 mmol) of triisopropyl borate was added and stirring was continued at the same temperature for 30 minutes. After that, the reaction mixture was allowed to slowly warm up to room temperature and stirred for 3 hours in the process. After completion of the reaction, 100 mL of 2 M hydrochloric acid was added to the mixture and stirred at room temperature for 2 hours. The reaction solution was separated and the aqueous phase was extracted with ethyl acetate. All organic phases were combined, washed with saturated sodium chloride solution, dried over anhydrous sodium sulfate and the solvent was subsequently evaporated. The residue was washed with dichloromethane in suspension to give 4.02 g of 6-hydroxy-2-naphthaleneboronic acid in 85% yield.