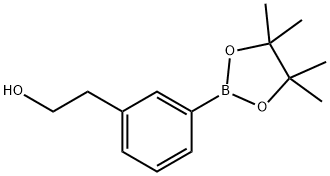
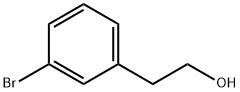
Stage 1: 3-bromophenethyl alcohol (3.0 g, 15 mmol), KOAc (4.39 g, 45 mmol), bis(pinacolato)diboron (5.68 g, 22 mmol), and PdCl2(dppf)2 (0.61 g, 0.74 mmol) were dissolved in anhydrous DMSO (30 mL). The reaction mixture was heated and stirred at 120 °C for 18 h under nitrogen protection. After the reaction was completed, it was cooled to room temperature and diluted by adding EtOAc (60 mL). The reaction mixture was filtered through Celite and the filter cake was washed with EtOAc (500 mL). The combined organic phases were washed sequentially with saturated NaHCO3 solution (150 mL), water (150 mL) and brine (150 mL), dried over MgSO4, filtered and concentrated under reduced pressure. The crude product was purified by automated column chromatography using EtOAc/heptane (gradient 0-100%) as eluent, and further purified by column chromatography (33% EtOAc/heptane) to afford 2-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)ethanol as a light yellow oil (3.30 g, 89% yield).1H NMR (300 MHz, CDCl3) δ (ppm): 7.70 (2H, m), 7.35 (2H, m), 3.89 (2H, t, J = 6.6 Hz), 2.90 (2H, t, J = 6.6 Hz), 1.51 (1H, s), 1.37 (12H, s).