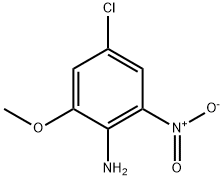
Step 2. Synthesis of 4-chloro-2-methoxy-6-nitroaniline. In a dry reaction flask, 2-amino-5-chloro-3-nitrophenol (752 mg, 4.0 mmol) and potassium carbonate (415 mg, 3.0 mmol) were dissolved in N,N-dimethylformamide (DMF). To the above mixture, iodomethane (625 mg, 4.4 mmol) was slowly added at room temperature. The reaction mixture was stirred continuously for 2 hours at room temperature. After completion of the reaction, the reaction was quenched with water and extracted with ethyl acetate (EtOAc). The organic layers were combined, washed with saturated brine and dried over anhydrous sodium sulfate. The solvent was removed by concentration under reduced pressure to give the crude product. The crude product was purified by fast column chromatography to afford the target compound 4-chloro-2-methoxy-6-nitroaniline (450 mg, 46.4% yield). The product was analyzed by LC-MS and showed m/z 203 ([M-H]+).