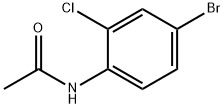
General procedure for the synthesis of 4-bromo-2-chloro-N-acetylaniline from N-(2-chlorophenyl)acetamide:
1. HBr (40% aqueous, 0.08 mL, 0.55 mmol) was added to a stirred suspension of N-(2-chlorophenyl)acetamide (75 mg, 0.5 mmol) and Selectfluor (213 mg, 0.6 mmol) in water (3.0 mL).
2. the mixture was stirred at room temperature for 5 minutes until the ingredients were completely consumed (monitored by TLC).
3. Upon completion of the reaction, the reaction was quenched with saturated Na2S2O3 aqueous solution (2.0 mL) and water (20.0 mL).
4. The organic phase was extracted three times with CH2Cl2 (10.0 mL) and combined.
5. After evaporation of the solvent, the residue obtained was purified by silica gel column chromatography (eluent: petroleum ether-ethyl acetate=6:1, v/v) to afford 4-bromo-2-chloro-N-acetylaniline as a white solid (108 mg, 95% yield).
[1] Chemistry - A European Journal, 2017, vol. 23, # 5, p. 1044 - 1047
[2] Tetrahedron Letters, 2016, vol. 57, # 48, p. 5390 - 5394
[3] Journal of Chemical Research, Synopses, 1997, # 11, p. 432 - 433
[4] European Journal of Organic Chemistry, 2018, vol. 2018, # 43, p. 5972 - 5979
[5] Gazzetta Chimica Italiana, 1908, vol. 38 II, p. 22