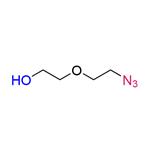
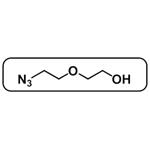
Add 50g of diethylene glycol and 34g of triethylamine to 500ml of dichloromethane with stirring, weigh 54g of p-toluenesulfonyl chloride dissolved in 150ml of dichloromethane, and then add it dropwise to the reaction system under the condition of ice-water bath, and after dropping, the reaction will take place at 20 for 12h. The TLC shows the end of the reaction, and then add 400ml of water to wash, separate the liquid, dry the organic phase with anhydrous sodium sulfate, spin dry and then purify 55g of intermediate A through column chromatography. The organic phase was dried with anhydrous sodium sulfate and spun dry, and then purified by column chromatography to obtain 55g of Intermediate A, yield: 90%. The NMR data were as follows: 1HNMR (400MHz, CDCl3): δ: 7.792 (d, J=8.4Hz, 2H); 7.350 (d, J=8.4Hz, 2H); 4.158 (t, J=4.4Hz, 2H); 3.702~3.567 (m, 6H); 2.818 (s, 1H); 2.435 (s, 3H).