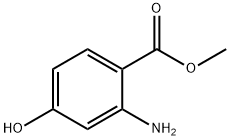
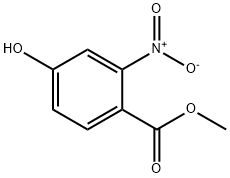
Step 3: Methyl 4-hydroxy-2-nitrobenzoate (4.410 g, 22.369 mmol) was dissolved in methanol (80 mL) under the protection of nitrogen atmosphere and stirred at room temperature. Ammonium formate (5.642 g, 89.475 mmol) and 5% Pd/C catalyst (450 mg) were subsequently added. After the reaction lasted for 5 h, the reaction mixture was filtered through a diatomaceous earth pad and washed well with methanol. The filtrate was concentrated under reduced pressure and the resulting residue was subjected to liquid-liquid partitioning with ethyl acetate (150 mL) and water (40 mL). After vigorous shaking, the organic layer was separated and washed sequentially with water (3 × 30 mL) and saturated saline (2 × 30 mL). The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give methyl 2-amino-4-hydroxybenzoate (3.593 g, 21.494 mmol, 96% yield) as an off-white solid with a melting point of 113-118 °C. The product was separated by 1H NMR (1H NMR, 1H NMR, 1H NMR, 1H NMR). The product was characterized by 1H NMR (DMSO-d6): δ 3.70 (s, 3H), 5.98 (dd, 1H), 6.10 (s, 1H), 6.59 (s, 1H), 7.54 (d, 1H), 9.81 (s, 1H); the mass spectrum [ES(-)] showed m/z 166 ([M-H]-).