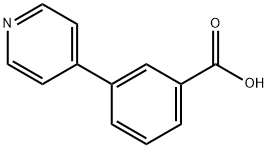
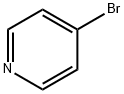
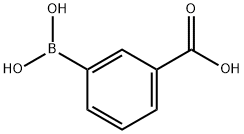
Example 165 Synthesis of A3-(pyridin-4-yl)benzoic acid: 3-Carboxyphenylboronic acid (1.5 g, 9 mmol) and 4-bromopyridine (1.5 g, 9.9 mmol) were dissolved in a mixed solvent of acetonitrile (40 mL) and water (40 mL), potassium carbonate (5.5 g, 40 mmol) and bis(triphenylphosphine)palladium(II) chloride (400 mg, 0.37 mmol). The reaction mixture was stirred under reflux conditions overnight. Upon completion of the reaction, the suspension was filtered while hot and the filtrate was concentrated to half the original volume. The concentrated aqueous phase was washed with dichloromethane. Subsequently, the aqueous phase was adjusted to pH=3 with 1 M hydrochloric acid, the solid was precipitated, filtered and the solid was washed with water. Finally, the solid residue was dried under vacuum to give 1.5 g of 3-(pyridin-4-yl)benzoic acid in 83% yield.LC-MS (ESI) m/z: 200(M+1)+.