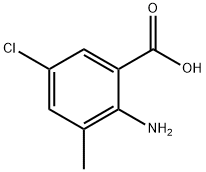
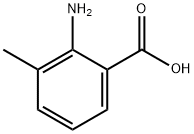
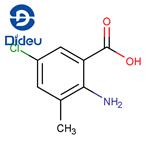
General procedure for the synthesis of 2-amino-5-chloro-3-methylbenzoic acid from 2-amino-3-methylbenzoic acid: In a 100 mL round-bottomed flask, 20 mL of 2-amino-3-methylbenzoic acid, 50 mL of N,N-dimethylformamide (DMF), and 30 mmol of N-chlorosuccinimide (or N-bromosuccinimide) were added. The mixture was stirred under reflux conditions for 3 hours. After the reaction was completed, the reaction solution was poured into ice water. The pH was adjusted to 6 with dilute hydrochloric acid and then filtered. The resulting solid was washed with a small amount of ethanol to give a gray solid 2-amino-5-halogeno-3-methylbenzoic acid (halogen was chlorine or bromine). The yield was 83%.
[1] Journal of Agricultural and Food Chemistry, 2012, vol. 60, # 44, p. 10942 - 10951
[2] Australian Journal of Chemistry, 2014, vol. 67, # 10, p. 1491 - 1503
[3] Patent: CN103450154, 2016, B. Location in patent: Paragraph 0063-0065
[4] Patent: CN104031026, 2017, B. Location in patent: Paragraph 0051; 0052; 0053; 0054
[5] Journal of Chemical Research, 2008, # 9, p. 530 - 533