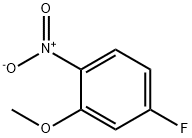
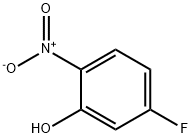
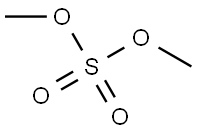
In a 5L four-necked flask, 2500 g of N,N-dimethylformamide (DMF), 5-fluoro-2-nitrophenol (500 g, 3.20 mol) and anhydrous potassium carbonate (483 g, 3.50 mol) were added and the mixture was heated up to 60 °C. Dimethyl sulfate (424 g, 3.36 mol) was slowly added dropwise for a controlled period of 2-3 hours. After the dropwise addition, the reaction temperature was raised to 90-100 °C and the reaction was maintained for 5-6 hours. The reaction progress was monitored by gas chromatography (GC) until the content of 5-fluoro-2-nitrophenol was below 0.5%. The reaction mixture was cooled to 50 °C, at which point the monomethyl potassium sulfate salt precipitated. The filtrate was subjected to decompression distillation to recover DMF, resulting in 2-nitro-5-fluoroanisole (515 g, yellow oily liquid). The product was analyzed by GC with 99.5% purity and 94.1% yield.
[1] Chemical and Pharmaceutical Bulletin, 1991, vol. 39, # 9, p. 2323 - 2332
[2] Patent: CN106083536, 2016, A. Location in patent: Paragraph 0035; 0036
[3] Heterocycles, 1992, vol. 34, # 12, p. 2301 - 2311
[4] Patent: WO2010/71885, 2010, A1. Location in patent: Page/Page column 443
[5] Patent: WO2010/115688, 2010, A1. Location in patent: Page/Page column 146