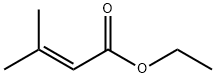
A suspension of 60% NaH in mineral oil (4.12 g, 0.11 mol, 1.2 eq.) in dry THF (100 mL) was cooled under nitrogen to 0C. A solution of triethyl acetate phosphate (22.1 mL, 0.11 mol, 1.3 eq.) in dry THF (30 mL) was then added. After stopping the bubbling, acetone (6.3 mL, 0.09 mol, 1 eq.) was added and the resulting mixture was heated to room temperature and stirred overnight. The mixture was quenched by the addition of water (100 mL) and then extracted with ether (2 x 100 mL). The combined organic extracts were washed with water (150 mL) followed by brine (150 mL). The organic layer was dried with anhydrous MgSO4 and the solvent was removed under vacuum. The crude product was purified using rapid chromatography (19:1 pentane:ether) to afford the pale yellow oily product ethyl 3-methyl-2-butenoate (9.67 g, 88%).