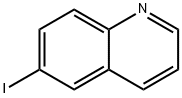
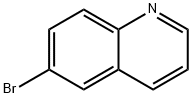
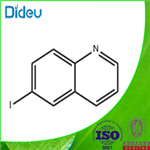
General procedure for the synthesis of 6-iodoquinoline from 6-bromoquinoline: sodium iodide (4.32 g, 28.8 mmol), copper(I) iodide (137 mg, 0.72 mmol), N,N'-dimethyl-cyclohexane-1,2-diamine (0.227 mL, 1.44 mmol) and 6-bromoquinoline (3 g, 14.4 mmol) in a solution of dioxane (15 mL). The reaction tube was flushed with nitrogen and sealed with a PTFE septum. Nitrogen was bubbled into the solution through a needle for 10 min to ensure that gas escaped through the needle. After removing the needle, the reaction mixture was stirred at 110°C for 15 hours. Upon completion of the reaction, the green suspension was cooled to room temperature, poured into ice water and extracted with dichloromethane. The organic layer was collected, dried with anhydrous magnesium sulfate, filtered and concentrated in vacuum. The crude product was purified by silica gel column chromatography, eluting sequentially with 100% dichloromethane and dichloromethane/methanol (95:5, v/v) to afford 3.56 g (97% yield) of 6-iodoquinoline as a light yellow solid.1H-NMR (DMSO-d6) δ: 8.93 (1H, dd, J = 1.5, 4.1 Hz), 8.47 (1H, d, J = 2.0 Hz ), 8.33 (1H, d, J = 8.6 Hz), 8.02 (1H, dd, J = 2.0, 8.6 Hz), 7.80 (1H, d, J = 8.6 Hz), 7.56 (1H, dd, J = 4.1, 8.6 Hz).