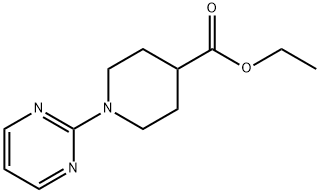
2-Chloropyrimidine (2.28 g, 20 mmol) and ethyl 4-piperidinecarboxylate (4.72 g, 30 mmol) were mixed in toluene (10 mL) and heated to reflux for 18 hours. After completion of the reaction, the mixture was cooled to room temperature, diluted with ethyl acetate and washed with saturated aqueous sodium bicarbonate. The organic layer was separated, dried and the solvent was removed under vacuum. The residue was purified by rapid chromatography on silica gel (eluent: ethyl acetate/petroleum ether, 40/60, v/v) to afford ethyl 1-(2-pyrimidinyl)piperidine-4-carboxylate as a colorless oil in a yield of 4.00 g in 81%. Mass spectrum (ESI+): [M + H]+ = 236.0.
[1] Farmaco, 1993, vol. 48, # 10, p. 1439 - 1445
[2] Patent: WO2010/97576, 2010, A1. Location in patent: Page/Page column 39
[3] Journal of Medicinal Chemistry, 2013, vol. 56, # 21, p. 8696 - 8711