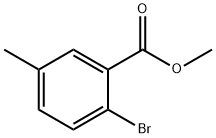
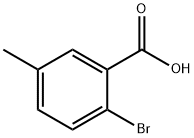
Step 1: Preparation of methyl 2-bromo-5-methylbenzoate
Oxalyl chloride (0.13 mL, 1.5 mmol) was slowly added to a mixed solution of dichloromethane (DCM, 4 mL) and N,N-dimethylformamide (DMF, 1 drop) of 2-bromo-5-methylbenzoic acid (215 mg, 1.0 mmol) at 0 °C. The reaction mixture was stirred at room temperature for 1 hour before oxalyl chloride (0.13 mL, 1.5 mmol) was added again. After continued stirring for 1 hour, methanol (MeOH, 2 mL) was added and the reaction mixture continued to stir for 6 hours. Upon completion of the reaction, the pH was adjusted to 9 with sodium carbonate (Na2CO3) solution and extracted with dichloromethane (DCM, 2 x 10 mL). The organic layers were combined, washed with saturated saline, dried over anhydrous sodium sulfate (Na2SO4), filtered and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography to afford methyl 2-bromo-5-methylbenzoate (210 mg, 92% yield) as a colorless oil using petroleum ether-ethyl acetate (PE-EA, 15:1) as eluent.
1H NMR (400 MHz, CDCl3): δ 7.60 (d, J = 1.6 Hz, 1H), 7.53 (d, J = 8.8 Hz, 1H), 7.15 (q, J = 3.2 Hz, 1H), 3.92 (s, 3H), 2.33 (s, 3H) ppm.