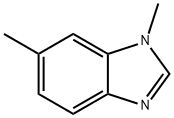
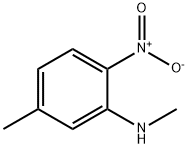
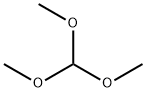
Step B: Preparation of 1,6-dimethyl-1H-benzo[d]imidazole: N,5-dimethyl-2-nitroaniline (20.2 g, 122 mmol), trimethyl orthoformate (133 mL, 1,216 mmol), 10% palladium carbon (6.47 g, 6.1 mmol) and p-toluenesulfonic acid monohydrate (2.31 g, 12.2 mmol) were dissolved in methanol ( 240 mL). The reaction system was placed under a hydrogen balloon at 1 atmosphere pressure and the reaction was stirred at room temperature for 5 hours. Upon completion of the reaction, the catalyst was removed by filtration and the filter cake was washed with methanol (200 mL). The filtrates were combined and concentrated under reduced pressure. The concentrated residue was treated with saturated sodium bicarbonate solution (50 mL) and ethyl acetate (300 mL) to separate the organic layer. The organic layer was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 1,6-dimethyl-1H-benzo[d]imidazole (17.7 g, quantitative yield) as a solid.