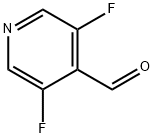
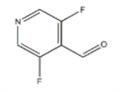
General procedure for the synthesis of 3,5-difluoropyridine-4-carbaldehyde from methyl formate and 3,5-difluoropyridine:
Preparation of Example 10: N-[(3,5-difluoro-4-pyridinyl)methyl]-2-(trifluoromethyl)pyridine-3-carboxamide (Compound A126)
Step 1: Synthesis of 3,5-difluoropyridine-4-carbaldehyde
A 2M solution of LDA (47.792 mL, 95.58 mmol) was diluted with 50 mL of THF at 0°C and cooled to -78°C. Subsequently, a 100 mL THF solution of 3,5-difluoropyridine (7.886 mL, 86.89 mmol) was added dropwise, keeping the temperature lower than -70°C (the dropwise addition process was controlled to be completed within 20 min). The reaction mixture formed a yellow suspension. Stirring was continued at -78°C for 3 hours. Then, 25 mL of THF solution of methyl formate (10.8 mL, 173.79 mmol) was added and the reaction mixture was transformed into a light yellow solution and stirred at -75°C for 45 minutes. The reaction mixture was transferred by cannula to 100 mL of stirred saturated aqueous NaHCO3 solution, maintained at about 0°C. The solution was extracted with EtOAc, the organic phases were combined and washed with brine and dried with 1N HCl. The solvent was evaporated at 165 mbar, 30°C to give 36.7 g of yellow liquid crude product. The crude product was purified by fast chromatography (eluent: CH2Cl2) to give a light yellow oil (7.85 g), which was crystallized after standing.
1H-NMR (CDCl3) data: δ 10.4 (s, 1H), 8.57 (s, 2H).