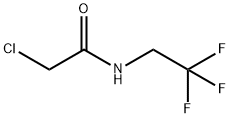
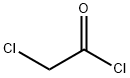
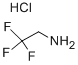GENERAL STEPS: 30.4 g (759.3 mmol) of sodium hydroxide and 50 mL of water were added to a 500 mL four-necked flask and stirred at room temperature until completely dissolved, then cooled to 5 °C. A solution was prepared by dissolving 50 g (370.4 mmol) of 2,2,2-trifluoroethylamine hydrochloride in 60 mL of water and added slowly and dropwise to the above sodium hydroxide solution at 5 °C. 85 mL of tert-butyl methyl ether was added and stirred continuously for 30 minutes. Subsequently, a solution of 43.9 g (388.9 mmol) of chloroacetyl chloride dissolved in 15 mL of tert-butyl methyl ether was added slowly dropwise while maintaining 5°C. The reaction mixture was heated to 10 °C and stirring was continued for 1 hour. After confirming complete consumption of 2,2,2-trifluoroethylamine by gas chromatography, the reaction mixture was warmed up to room temperature and partitioned. The aqueous layer was extracted with 100 mL of tert-butyl methyl ether, all organic layers were combined, and the solvent was concentrated by distillation under reduced pressure to give a tert-butyl methyl ether solution of 2-chloro-N-(2,2,2-trifluoroethyl)acetamide. The product was analyzed by high performance liquid chromatography with internal standard method to give 65 g of product in 100% yield and a solution concentration of 25.1% by weight. The solution was added dropwise to heptane to induce crystallization, and the melting point of the resulting 2-chloro-N-(2,2,2-trifluoroethyl)acetamide was 53.8 °C (as determined by DSC).