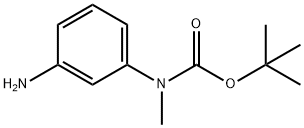
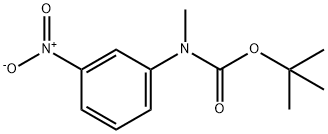
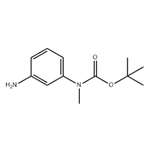
General procedure for the synthesis of 3-(N-tert-butoxycarbonyl-N-methylamino)aniline from tert-butyl methyl(3-nitrophenyl)carbamate: the product of step b (3.30 g, 13.1 mmol), 10% palladium-carbon catalyst (300 mg), and a THF-MeOH solvent mixture (1:1, 50 mL) were added to a round-bottom flask. The system was evacuated and displaced three times with hydrogen. The reaction mixture was stirred vigorously overnight under hydrogen atmosphere. Upon completion of the reaction, the catalyst was removed by filtration through a diatomaceous earth pad and the filtrate was concentrated under reduced pressure to give a white solid product (2.90 g, 99% yield). The structure of the product was confirmed by 1H NMR (CDCl3): δ 7.10 (1H, t), 6.62 (2H, m), 6.50 (1H, m), 3.66 (2H, br s), 3.22 (3H, s), 1.46 (9H, s).