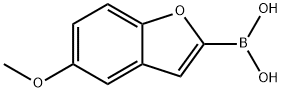
Step A: 5-methoxybenzofuran (1 g, 6.8 mmol) was dissolved in anhydrous tetrahydrofuran (10 mL) and cooled to -30 °C under nitrogen protection. n-Butyllithium (3.5 mL, 8.9 mmol, 2.5 M hexane solution) was added slowly dropwise, keeping the internal temperature of the reaction system below -30 °C. After the dropwise addition, stirring was continued for 30 min, and the solution was red. After maintaining the reaction at -30 °C for 1 h, trimethyl borate (1 mL, 8.8 mmol) was added slowly over a period of 10 min, and a change in the color of the solution to light brown was observed. Subsequently, the reaction system was slowly warmed up to 10 °C over a period of 2 hours. Upon completion of the reaction, the reaction was quenched with 6 M hydrochloric acid (10 mL) and extracted with ethyl acetate (30 mL). The organic phases were combined, washed sequentially with water (30 mL) and saturated saline (30 mL) and dried over anhydrous magnesium sulfate. The desiccant was removed by filtration, the organic phase was concentrated to dryness under reduced pressure, and the product was induced to precipitate by the addition of hexane. The precipitate was collected by filtration and washed with cold hexane to give 5-methoxybenzofuran-2-boronic acid (983 mg, 76% yield) as an off-white solid. The product was characterized by 1H NMR (500 MHz, DMSO-d6): δ 8.53 (broad single peak, 2H), 7.47 (broad double peak, J=8.8 Hz, 1H), 7.20 (broad single peak, 1H), 6.94 (broad double-double peak, J=8.8,2.5 Hz, 1H), 3.79 (broad single peak, 3H).