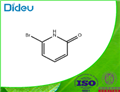
General procedure for the synthesis of 2-bromo-6-hydroxypyridine from 2,6-dibromopyridine: 2,6-dibromopyridine (4 g, 17 mmol), potassium tert-butoxide (20 g, 0.27 mol), and redistilled tert-butanol (100 mL) were mixed and reacted at reflux overnight. After completion of the reaction, the mixture was cooled and the solvent was removed under vacuum. Subsequently, the ice-water mixture was carefully added and the aqueous layer was extracted with chloroform (100 mL x 2) to remove unreacted starting material. Next, the aqueous layer was acidified with 3N hydrochloric acid and again extracted with chloroform (100 mL x 2). The organic phases were combined, washed with brine, dried over anhydrous sodium sulfate and concentrated to give pure 2-bromo-6-hydroxypyridine (2.5 g, 85% yield) as a white solid.