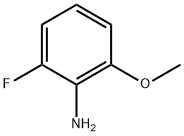
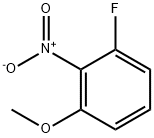
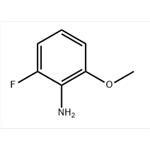
General procedure for the synthesis of 2-fluoro-6-methoxyaniline using 3-fluoro-2-nitroanisole as starting material: 10% Pd/C catalyst (7.46 g, 70.1 mmol) and ethanol (400 mL) were added to a Parr reactor followed by 1-fluoro-3-methoxy-2-nitrobenzene (40.0 g, 234 mmol). The reactor was evacuated and filled with hydrogen (50 psi) and the reaction was shaken for 5 h under hydrogen atmosphere. After completion of the reaction, the catalyst was removed by Celite filtration and the filtrate was concentrated to give 2-fluoro-6-methoxyaniline (30.0 g, 83% yield) as a colorless oil. The product was characterized by 1H NMR (400 MHz, CDCl3): δ 3.75 (broad single peak, 2H), 3.86 (single peak, 3H), 6.58-6.71 (multiple peaks, 3H); the molecular ion peak [M + H]+ = 142 was detected by LC-MS (LC-ES).
[1] Patent: WO2018/69863, 2018, A1. Location in patent: Page/Page column 248
[2] Journal of the American Chemical Society, 1932, vol. 54, p. 2973,2976
[3] Journal of the Chemical Society, 1931, p. 981
[4] Journal of Medicinal Chemistry, 1993, vol. 36, # 11, p. 1641 - 1653