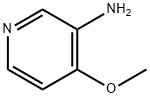
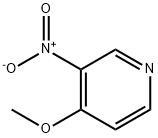
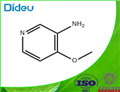
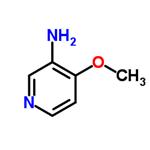
Example 93: Synthesis of 4-hydroxy-4-[3-(trifluoromethyl)phenyl]piperidine-1-carboxylic acid (7-methoxy-thiazolo[5,4-b]pyridin-2-yl)-amide; Step 1: To a solution of 4-methoxy-3-nitropyridine (5.0 g, 32.44 mmol) in ethanol (100 mL) was added 10% palladium/carbon catalyst (200 mg). The reaction mixture was placed under hydrogen atmosphere (50 psi) and the reaction was shaken at room temperature for 6 hours. The progress of the reaction was monitored by thin layer chromatography (TLC, 50% ethyl acetate/hexane) to confirm complete conversion of the feedstock. Upon completion of the reaction, the catalyst was removed by diatomaceous earth filtration and the filtrate was concentrated under reduced pressure to afford 3-amino-4-methoxypyridine (4.0 g, 32.44 mmol, 100% yield), the product was a dark red oil.
[1] Patent: US2007/270433, 2007, A1. Location in patent: Page/Page column 47
[2] Patent: WO2012/67664, 2012, A1. Location in patent: Page/Page column 53
[3] Inorganic Chemistry, 2013, vol. 52, # 7, p. 3653 - 3662
[4] Patent: US2011/9405, 2011, A1. Location in patent: Page/Page column 49-50
[5] Chemistry - A European Journal, 2012, vol. 18, # 51, p. 16358 - 16368