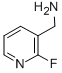
General procedure for the synthesis of 2-fluoro-3-pyridinylmethylamine from 2-fluoro-3-cyanopyridine: first, C-(2-fluoro-pyridin-3-yl)-methylamine hydrochloride was prepared. Concentrated hydrochloric acid (0.46 mL) was slowly added to a suspension of methanol (10 mL) containing 2-fluoro-3-cyanopyridine (0.34 g, 2.8 mmol) and 5% Pd/C (0.5 g) at room temperature. Subsequently, the reaction suspension was stirred continuously for 6 hours under a hydrogen atmosphere at 1 atmosphere. Upon completion of the reaction, the reaction mixture was filtered and the filtrate concentrated. Ether was added to the concentrated residue and HCl gas was passed through the suspension bulge. The resulting precipitate was filtered and dried to give the target product 2-fluoro-3-pyridylmethylmethylamine (0.37 g, 82% yield). Substance spectral analysis (ES) of the product showed a m/z of 127.1 ([M + H]+). The NMR hydrogen spectrum (400 MHz, DMSO-d6) data were as follows: δ 8.65 (broad single peak, 3H), 8.24 (multiple peaks, 1H), 8.16 (multiple peaks, 1H), 7.41 (multiple peaks, 1H), 4.06 (multiple peaks, 2H).