General Steps:
Example 1; Synthesis of 3-methyl-6-(4-(methylsulfonyl)phenyl)-N-phenylpiperazine-2-carboxamide (Compound IV-1) Method A:
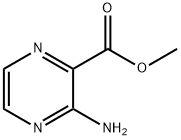
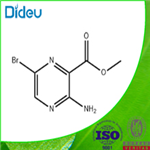
Step 1: Preparation of methyl 3-amino-6-bromopyrazine-2-carboxylate[A
Methyl 3-aminopyrazine-2-carboxylate (8.35 g, 54.53 mmol) was mixed with N-bromosuccinimide (9.705 g, 54.53 mmol) in acetonitrile (100 mL), and the reaction was stirred for 16 hours at room temperature. After completion of the reaction, the precipitate was collected by filtration, washed with acetonitrile and dried to afford the target product methyl 3-amino-6-bromopyrazine-2-carboxylate as a yellow solid (11.68 g, 92% yield).
1H NMR (400.0 MHz, DMSO-d6) δ 3.85 (s, 3H), 7.55 (br s, 2H), 8.42 (s, 1H) ppm; MS (ES+) m/z 233.