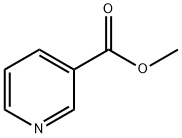
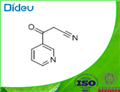
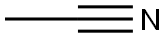To a stirred solution of methyl nicotinate (XVIII; 8 g; 58 mmol) in toluene (110 mL) at 0 °C was slowly added sodium hydride (2.8 g; 110 mmol). The reaction mixture was kept stirred at 0 °C for 30 min, followed by dropwise addition of acetonitrile (12 g; 91 mmol). The reaction mixture was heated to reflux for 72 hours. After completion of the reaction, the mixture was cooled, concentrated under reduced pressure and diluted with ice-cold water. The pH of the reaction mixture was adjusted to acidic with glacial acetic acid. Subsequently, the aqueous layer was extracted with ethyl acetate (3 x 100 mL). The organic layers were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 3-oxo-3-(pyridin-3-yl)propanenitrile (XIX; 6 g, 70% yield) as a yellow solid. Mass spectrometry analysis showed [M-1] peak at 145.01.