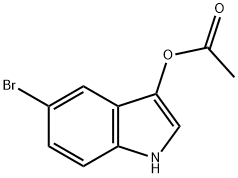
Method I.
The 3-hydroxyindole acetate, N-bromosuccinimide and 150 mL of carbon tetrachloride. It was added into 250 mL round bottom flask and refluxed in oil bath for 2 h. The reaction process was monitored by TLC. At the completion of the reaction, the solution changed from dark yellow to light yellow and a white solid (succinimide) was precipitated. The filtrate was withdrawn and washed, and the filtrate was collected and evaporated to obtain 5-bromo-3-hydroxyindole acetate.
Method II.
In a 500 mL single-necked flask, 3-hydroxyindole acetic acid ester was dissolved in tetrahydrofuran solution, N-bromosuccinimide was added in an ice bath, and the reaction was stirred at 0C for 3 h. The reaction was completed. The reaction mixture was poured into 150 mL of ice water and extracted with methyl tert-butyl ether (100 mL4), the organic phases were combined, dried with anhydrous sodium sulfate, and concentrated to obtain 5-bromo-3-hydroxyindole acetate.