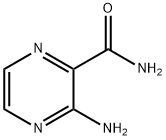
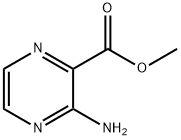
Example 2 Synthesis of 3-aminopyrazine-2-carboxamide (Intermediate 1): methyl 3-aminopyrazine-2-carboxylate (5.0 g, 33 mmol) was dissolved in concentrated ammonium hydroxide solution (30 mL), heated to 60°C and kept for 3 hours. After completion of the reaction, the mixture was cooled to room temperature and the precipitated solid was collected by filtration. The solid was washed well with water and ether in turn to afford the title compound 3-aminopyrazine-2-carboxamide as a brown solid (3.7 g, 82% yield). The product was characterized by 1H NMR (500 MHz, DMSO-d6): δ 8.19 (d, J = 2.4 Hz, 1H), 8.07 (br s, 2H), 7.80 (d, J = 2.4 Hz, 1H), 7.59 (br s, 2H). Mass spectrum (ES+): m/z 139 ([M + H]+).