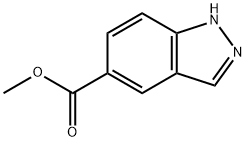
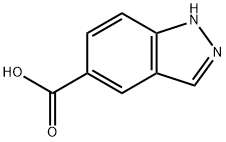
Concentrated sulfuric acid (0.2 mL) was slowly added to a methanolic (5 mL) suspension of 1H-indazole-5-carboxylic acid (470 mg, 2.9 mmol). The reaction mixture was heated to 70 °C and the reaction was stirred at this temperature overnight. Upon completion of the reaction, the mixture was cooled to room temperature and diluted and neutralized by adding water (10 mL), saturated aqueous sodium bicarbonate (5 mL), and ethyl acetate (30 mL) in that order. The organic and aqueous phases were separated and the aqueous phase was extracted once more with ethyl acetate (30 mL). All organic phases were combined, dried with anhydrous sodium sulfate, filtered and concentrated to dryness under reduced pressure to afford methyl 1H-indazole-5-carboxylate (466 mg, 2.65 mmol, 88% yield) as a light pink to yellow solid. Mass spectrometry analysis showed that the measured value of C9H8N2O2 was (M + H)+ 176.9.
[1] European Journal of Medicinal Chemistry, 2017, vol. 127, p. 470 - 492
[2] Patent: WO2013/78237, 2013, A1. Location in patent: Page/Page column 81; 82
[3] Patent: WO2016/196776, 2016, A2. Location in patent: Paragraph 00725
[4] Patent: WO2013/96496, 2013, A2. Location in patent: Page/Page column 26