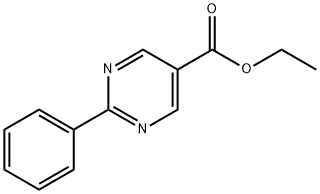
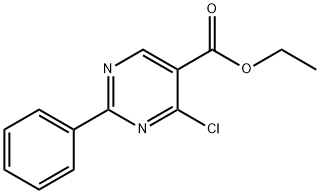
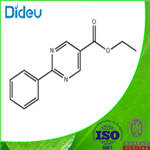
General procedure for the synthesis of ethyl 2-phenylpyrimidine-5-carboxylate from ethyl 4-chloro-2-phenylpyrimidine-5-carboxylate: Under argon protection, anhydrous ethanol (5 mL), sodium metal (0.426 g, 18.5 mmol), benzylcarbamidine hydrochloride (2.90 g, 18.5 mmol), and diethyl 2-(ethoxymethylene)malonate were added sequentially to a round-bottomed flask ( 4.0 g, 18.5 mmol). The reaction mixture was stirred at room temperature for 9 h and subsequently poured into ice water. The precipitate was collected by filtration, washed with cold water and recrystallized in a solvent mixture of EtOH-MeOH-CHCl3 (1:1:1, 45 mL) to afford ethyl 4-hydroxy-2-phenylpyrimidine-5-carboxylate (8) as a white crystalline solid in 50% yield. Compound 8 (1.2 g, 4.91 mmol) was refluxed with POCl3 (9.8 g, 63.9 mmol) at 100 °C for 1 hour. Upon completion of the reaction, the excess POCl3 was removed in vacuum, the reaction mixture was poured into ice water, filtered and washed with cold water to afford chloride 9 as a white amorphous solid in 98% yield. Subsequently, chloride 9 was stirred with zinc powder (0.176 g, 2.69 mmol, 4 eq.) in anhydrous THF (2 mL) at 60 °C for 1 h. Then 5 drops of acetic acid were added. After continued stirring at 60 °C for 23 h, the reaction mixture was cooled to room temperature, CH2Cl2 (3 mL) was added, filtered and the solvent was evaporated. Purification by silica gel column chromatography (n-hexane-EtOAc, 0.6%) afforded ethyl 2-phenylpyrimidine-5-carboxylate as a white crystalline solid in 50% yield.