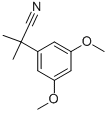
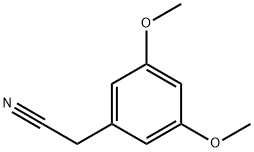
Under argon protection, 3,5-dimethoxyphenylacetonitrile (10 g, 56.43 mmol) was dissolved in anhydrous DMF (280 mL) with iodomethane (10.5 mL) and added dropwise through a cannula to a suspension of sodium hydride (4.06 g, 169.20 mmol) in DMF (30 mL) that was pre-cooled to 0 °C. After dropwise addition, the reaction mixture was slowly warmed to room temperature and stirring was continued for 1.5 hours. Upon completion of the reaction, the reaction was quenched with saturated aqueous ammonium chloride solution and subsequently diluted with ether. The organic layer was separated and the aqueous phase was further extracted with ether. All organic layers were combined, washed with brine, dried over anhydrous magnesium sulfate and concentrated under reduced pressure to remove the solvent. The residue was purified by fast column chromatography on silica gel, the eluent being a hexane solution of 10-30% ethyl acetate to afford the target product 2-(3,5-dimethoxyphenyl)-2-methylpropanenitrile (11.3 g, 98% yield) as a viscous oil.1H NMR (500 MHz, CDCl3) δ 6.61 (d, J=2.0Hz, 2H, ArH), δ 6.27 (t, J=2.0Hz, 1H, ArH), 3.81 (s, 6H, OCH3), 1.70 (s, 6H, C(CH3)2).