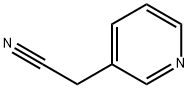
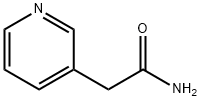
The general procedure for synthesizing 3-pyridylacetonitrile from pyridine-3-acetamide is as follows: general method: after completing the preparation of the amide intermediate, the operation of Example A is carried out. Decide the state of the reaction vessel according to the relationship between the boiling point of the amide intermediate at atmospheric pressure and the reaction temperature TB: if the boiling point of the amide intermediate is equal to or lower than TB, close the reaction vessel; if higher than TB, keep the reaction vessel open. Start the stirrer, set the speed to 600 rpm, adjust the reaction temperature to TB and maintain this temperature for TD hours until the reaction is essentially complete. Subsequently, the reaction vessel was sealed and connected to a vacuum pump, the system vacuum was adjusted to 20-50 mbar (depending on the nature of the nitrile product), and the distillate was collected as the target nitrile product. The yield of the product was calculated and samples were taken for NMR and elemental analysis to characterize the structure and purity of the resulting nitrile product. The specific reaction conditions and characterization results are detailed in Tables A-7 to A-12 below. the characterization data confirmed that the resulting nitrile products were of very high purity (>99%). In this series of examples for the preparation of the nitrile product, it is recommended to add 10 g of phosphorus pentoxide as a catalyst at once at the beginning of the reaction.