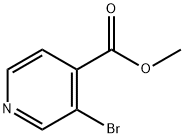
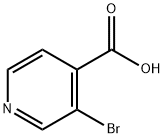
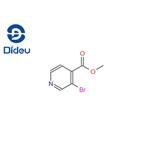
3.98 g (20 mmol, 1 eq.) of 3-bromoisonicotinic acid was dissolved in 50 ml of methanol and 4 ml of concentrated sulfuric acid was added as catalyst. The reaction mixture was heated to reflux. After completion of the reaction, it was cooled to room temperature and extracted three times with ethyl acetate. The organic phases were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. 2.65 g (62% yield) of methyl 3-bromoisonicotinate was obtained. The structure of the product was confirmed by 1H NMR (CDCl3): δ 4.02 (s, 3H), 7.64 (d, J = 4.9 Hz, 1H), 8.63 (d, J = 4.9 Hz, 1H), 8.88 (s, 1H).