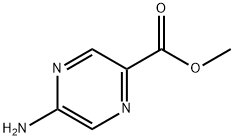
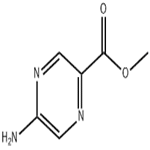
General procedure for the synthesis of methyl 5-aminopyrazine-2-carboxylate from methyl 5-chloropyrazine-2-carboxylate: methyl 5-chloropyrazine-2-carboxylate (2 g, 0.0115 mol) was dissolved in 80 mL of dimethyl sulfoxide (DMSO). Sodium azide (3 g, 0.0463 mol) and triphenylphosphine (4.6 g, 0.01738 mol) were added sequentially to the solution, and the reaction mixture was refluxed at 120 °C for 4 hours. After the reaction was completed, 20 mL of 1 N hydrochloric acid solution was added and the reaction was continued at 120 °C for 2 hours. The reaction mixture was cooled to room temperature and neutralized with 90% aqueous sodium bicarbonate solution, followed by extraction of the product with ethyl acetate. The organic phase was dried with anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The residue was washed with n-pentane to give 0.7 g (39.5% yield) of the target compound 5-aminopyrazine-2-carboxylic acid methyl ester as a yellow solid. The product was characterized by 1H NMR (400 MHz, DMSO-d6): δ 8.53 (d, J = 1.2 Hz, 1H), 7.91 (d, J = 1.2 Hz, 1H), 7.39 (s, 2H), 3.79 (s, 3H). Elemental analysis (C6H7N3O2) calculated value: [M] 153.14; mass spectrometry (ESI) m/z: [M-H]+ 152.05.