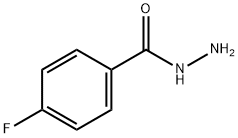
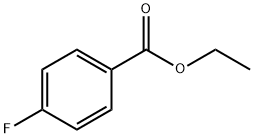
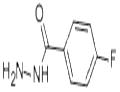
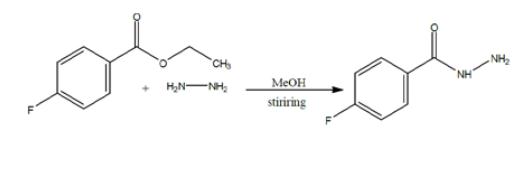
The synthesized ester (12.5ml) was taken into a round bottom flask (250ml), 50ml of absolute ethanol & 37.5ml of hydrazine hydrate was added in the flask. The ester, ethanol & hydrazine hydrate for the synthesis of hydrazide must be in following ratio: Ester: Absolute ethanol: Hydrazine hydrate 1 : 4 : 3 Then round bottom flask was covered with aluminium foil, placed on hot plate & allowed to stir it for almost 12-15 hrs. As the solid product appeared, reaction completion was checked by performing TLC using n-hexane, ethyl acetate & TLC cards & observed under UV lamp. After completion of reaction n-hexane was added. Solution filtered as needle like crystals appeared. Allowed it to dry & a shiny off white product was obtained that was hydrazide & product was calculated (79%).
Used in the stnthesis of fluorinated poly (1, 3, 4-oxadiazole-ether-imide). N-(3,5-dinitrobenzoyl)-N-(4-fluorobenzoyl)-hydrazine with 4-Fluorobenzhydrazide as solvent in NMP.
General procedure for the synthesis of 4-fluorobenzohydrazide from ethyl 4-fluorobenzoate: To a stirred solution of ethyl 4-fluorobenzoate (4.5 g, 26.7 mmol) in ethanol (30 mL) was added hydrazine hydrate (6.67 g, 133.5 mmol) at room temperature. The reaction mixture was heated to 85 °C and refluxed for 10 hours. Upon completion of the reaction, the mixture was cooled to room temperature and subsequently concentrated under reduced pressure. The resulting solid was washed with hexane (50 mL) and dried to give 4-fluorobenzhydrazide (4.2 g, quantitative yield). The structure of the product was confirmed by 1H NMR (400 MHz, DMSO-d6): δ 9.85 (s, 1H), 7.86 (dd, 2H), 7.25 (t, 3H), 4.46 (s, 1H), 3.47 (br s, 1H).
[1] Patent: WO2013/49559, 2013, A1. Location in patent: Page/Page column 92
[2] Bioorganic and Medicinal Chemistry, 2015, vol. 23, # 17, p. 6014 - 6024
[3] Patent: WO2011/52516, 2011, A1. Location in patent: Page/Page column 59
[4] Journal of Nanoscience and Nanotechnology, 2013, vol. 13, # 5, p. 3321 - 3330
[5] European Journal of Medicinal Chemistry, 2010, vol. 45, # 9, p. 3943 - 3949